Topic "Molecular Engineering"
A few words
Expertise in the engineering of redox membrane proteins
The I2M team is also recognized for its know-how in the field of protein engineering, in particular cytochromes P450 and their electron donors. We have developed original approaches for the creation of combinatorial libraries and functional mapping of modified enzymes. The engineering methods we use target either individual enzymes (modification of natural enzyme activities, engineering of active or recognition sites) or macromolecular assemblies (formation of redox complexes, formation of the shell of bacterial microcompartments, assemblies of individual enzymes). Molecular engineering is perfectly integrated with the metabolic engineering theme because both pursue the same objective: to obtain efficient synthetic metabolic systems. The enzymatic models used are oxidases (cytochromes P450), terpene synthetases and enzymes of the carotenoid production pathway (desaturases, cyclases, dioxygenases, etc.).
Ongoing projects
Project
Bacterial Microcompartments
La synthèse continue exige le développement de nouveaux outils biotechnologiques pour améliorer l’efficacité des cascades métaboliques créées dans des micro-organismes donnés. Deux stratégies majeures pour atteindre cet objectif sont la colocalisation spatiale des enzymes conçues et l’optimisation de leur orientation relative. Dans ce sens, notre première intention est d’apprendre de la nature, en contribuant à notre compréhension de la fonction des micro-compartiments bactériens (BMC). Les BMC sont des assemblages macromoléculaires qui encapsulent naturellement des enzymes qui participent à divers processus métaboliques. Nous étudions par divers moyens biophysiques le mécanisme qui conduit à l’assemblage de la coquille des BMC. L’objectif du second axe de ce projet est d’établir la composition précise des unités d’assemblage naturelles pour ensuite concevoir de nouvelles plateformes hexamériques.
Project
Combination of linker sequences on the activity of artificial fusion proteins.
Project
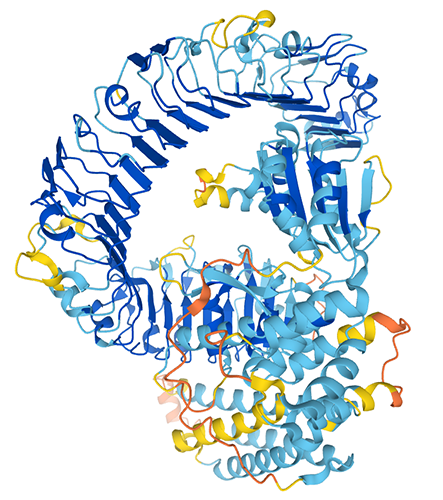
Interdomain dynamics in a multidomain oxidoreductase
Dans l’organisme, les médicaments subissent un métabolisme spécifique qui s’effectue principalement par les enzymes P450. Une seule flavoenzyme, la NADPH-cytochrome P450 réductase (CPR), donne les électrons nécessaires aux P450s pour leur catalyse. Jusqu’à présent, la plupart des variations interindividuelles de ce métabolisme ont été étudiées sur les polymorphismes du P450. Les modifications de l’équilibre conformationnel de la CPR, causées soit par des mutations, soit par l’interaction avec de petites molécules, peuvent augmenter son affinité pour un P450 spécifique, au détriment des autres. L’objectif de ce projet est de déchiffrer les facteurs (membrane du reticulum endoplasmique, état d’oxydation et variantes polymorphes de la CPR) qui contrôlent les modifications structurales et la dynamique de la CPR ancrée dans la membrane et leur implication dans le métabolisme des médicaments et les déficiences fonctionnelles potentielles. Ce projet vise à fournir de nouveaux outils pour décrypter les facteurs moléculaires modulant le paysage dynamique de la CPR et la spécificité des interactions dans les complexes CPR/P450.
Projet AlphaFold2
AlphaFold and protein complexes
AlphaFold2, an AI deep-learning system developed by DeepMind surpasses in accuracy all previous computing attempts for protein structure predictions. Recent versions were successfully extended to modeling of protein complexes including in some instances the potential to address large scale conformational changes involved in catalysis. DeepMind and EMBL’s European Bioinformatics Institute (EMBL-EBI) have partnered to create AlphaFold Protein Structure DataBase (ADB) that covers the complete human and 47 other key organisms (mouse, yeast, Arabidopsis, etc.), as well as the manually curated UniProt database. As of April 2022, AlphaFold DB provides open access to 992,316 protein 3D structure entries, most of them with no experimental structure available. We are using AlphaFold2_advanced version to look at the structures of complexes between P450 enzymes and P450-associated redox proteins and to their alternative geometries.
