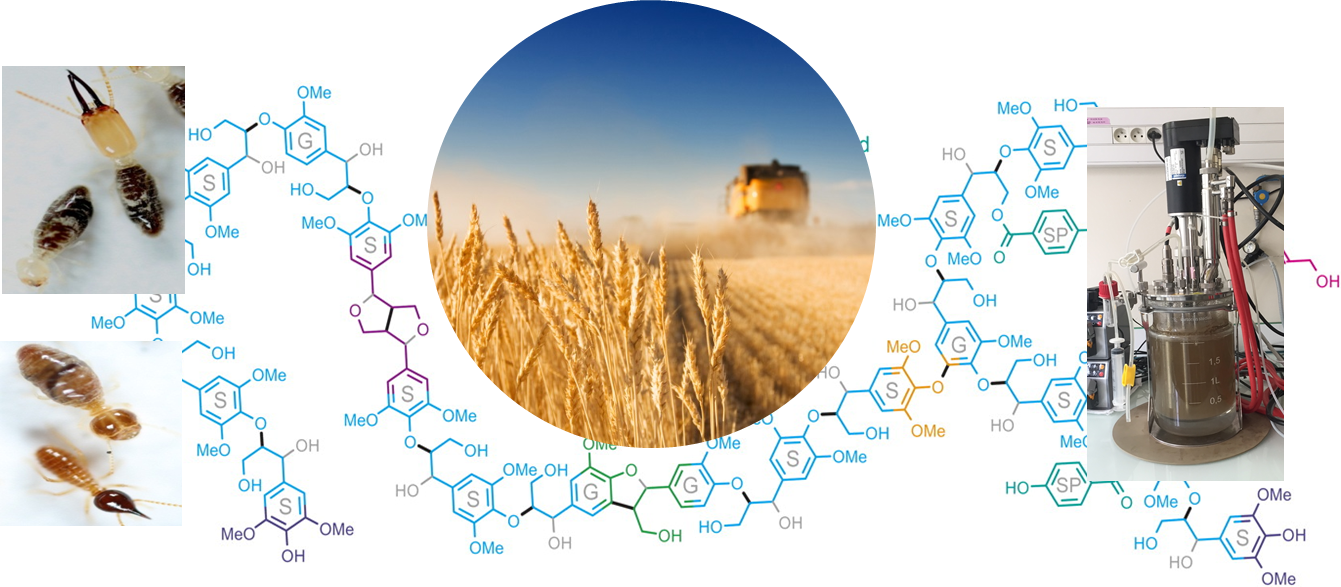
In current biorefineries, lignin is regarded as a by-product with little added value, whereas it could be considered as a renewable source of aromatic compounds for the production of biofuels and chemical compounds. The development of efficient lignin deconstruction systems would enable existing biorefinery processes to be improved and lignin to be valorized. Among lignin conversion routes, bioconversion by microbial species offers the advantage of being part of a green chemistry approach: low energy consumption, absence of solvents and toxic chemical reagents. As part of Louison Dumond's thesis, we highlighted the potential of microbial consortia derived from the digestive system of termites to degrade lignin. To achieve this, we combined bioreactor studies with cutting-edge analytical chemistry techniques and shotgun metagenomic and functional screening analyses. Our results highlighted the functional complementarity between the microbial species present in the microbial consortia and identified genetic targets involved in lignin deconstruction.
Background and challenges
Aujourd’hui, nos sociétés sont conscientes que les ressources fossiles telles que le pétrole, le gaz naturel ou le charbon sont limitées et que leur utilisation intensive a un impact important sur le climat. Cela nous amène à essayer de trouver de nouvelles façons de fournir à l’industrie de l’énergie et des molécules et produits chimiques de base. Dans ce cadre, la biomasse lignocellulosique peut être une source notable. Parmi les polymères constituant la lignocellulose, la lignine est sans doute la plus difficile à convertir en produits de valeur. Cependant, la gamme de molécules aromatiques qui peuvent être obtenues par la conversion de la lignine fait que la mise en place d’une bioraffinerie de lignine vaut la peine de relever le défi de sa dégradation.
Les termites sont connus comme les dégradateurs de lignocellulose les plus efficaces dans la nature ; ils peuvent se nourrir des polysaccharides de la lignocellulose et les transformer en énergie et en biomasse de termites. Néanmoins, le rôle exact du microbiome intestinal du termite dans la conversion de la lignine, n’a pas été entièrement caractérisé. Ainsi, nous avons entrepris d’étudier la capacité du microbiome digestif des termites à dégrader la lignine.
Results
Our studies in controlled bioreactors, combined with cutting-edge analytical chemistry techniques (HSQC NMR and 13C-Py-GC-MS), revealed the degradation of over 20% of the lignin present in the initial biomass, as well as structural modifications to the polymer with the appearance of metabolites indicative of lignin degradation. Shotgun metagenomic sequencing of the entire DNA of the microbial consortium enriched in the bioreactors has enabled us to reconstruct the genomes of the species present in the lignolytic microbial consortium and to identify the functional complementarities existing between the members of the community. Our results provide new insights into the underlying cooperation between bacteria to degrade lignocellulose and lignin.
Analysis of sequencing data coupled with functional screening metagenomics and enzyme activity measurements enabled us to identify genes encoding functions linked to plant wall deconstruction.
Our research has yielded microbial consortia capable of degrading lignin and new sequencing data for screening new enzymes of use to the biorefinery.
Outlook
These results open up broad prospects for the use of microbial consortia and their enzymes to modify and degrade lignin. We will complete the analysis of our sequencing data to identify new enzymes of interest for the biorefinery.
References
Résultats présentés à « Lignin Gordon Research Conferences ; publication en cours de rédaction.
Partnerships
IJPB – AgroParisTech, Wageningen University & Research, RISH Kyoto University, Warwick University, IRD, Université Paris 13–Sorbonne Paris Cité.
Contact
Guillermina Hernandez Raquet (hernandg@insa-toulouse.fr)